BRAND MANUFACTURERS
Chargeback ProcessingEnsure cost-effective reconciliation of low volumes of chargebacks. Use a robust processing solution when volume grows.
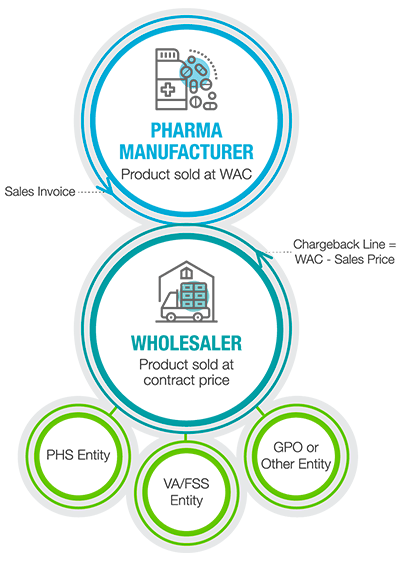
When a wholesaler distributes products through to an entity that has a direct contract with the pharma manufacturer for a price lower than WAC, a chargeback invoice will be generated by the wholesaler and submitted back to the manufacturer for verification and credit on the wholesaler’s normal invoice from the manufacturer.
Download a summary of our services.
How we support chargeback processing needs.
Prescription Analytics reconciles the invoiced lines against contracts that may be eligible and the specific pricing terms of the agreement to verify if the chargeback is legitimate. This makes a great deal of sense if the chargeback volumes are minimal, as the cost of implementing a full chargeback solution can be significant. We will work with you to assess volumes. If they increase significantly, we can introduce a best-in-class chargeback processing solution to support the larger volumes.
Services provided for chargeback support.
When chargeback volumes increase to the point where you need a full software solution, we will implement a more robust system. This strategy is is an effective way to control the higher fixed costs associated with chargeback processing. Both of our methodologies will include:
Continuous customer and contract setup and maintenance
Processing and verification of all submitted line items
Account maintenance and membership eligibility
Issue resolution with trading partners
Oversight and reporting by specific contract
Full data integration with client reporting system
Learn About Our Other Brand Services.
Click on any link to discover the many other ways we can help you make the smartest possible strategic and tactical business decisions.
